Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (16): 2588-2593.doi: 10.3969/j.issn.2095-4344.2014.16.021
Previous Articles Next Articles
Biomaterials for repair of sport-induced meniscal injuries: actuality and future
Wang Shi-zhe
- Department of Physical Education, Lanzhou University of Finance and Economics, Lanzhou 730020, Gansu Province, China
-
Revised:2014-02-16Online:2014-04-16Published:2014-04-16 -
Contact:Wang Shi-zhe, Department of Physical Education, Lanzhou University of Finance and Economics, Lanzhou 730020, Gansu Province, China -
About author:Wang Shi-zhe, Associate professor, Department of Physical Education, Lanzhou University of Finance and Economics, Lanzhou 730020, Gansu Province, China
CLC Number:
Cite this article
Wang Shi-zhe. Biomaterials for repair of sport-induced meniscal injuries: actuality and future[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2588-2593.
share this article

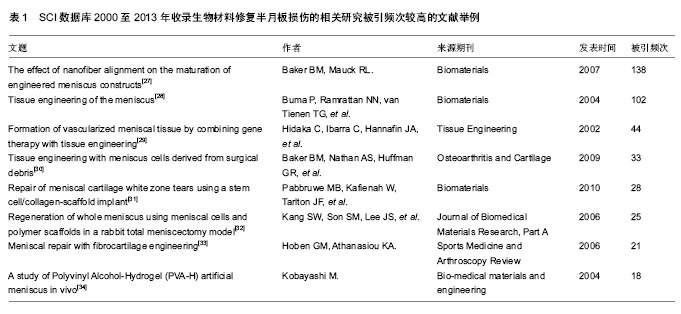
2.1 半月板的解剖结构和功能特征 解剖学中,膝关节呈现出一个半月形的形状,在胫骨平台关节面上,半月板在膝关节中呈现出凸型,在膝关节中起着缓冲、承载以及润滑的作用。半月板位于股骨内外侧髁与胫骨内侧髁的关节面之间,主要由纤维软骨构成,分内、外侧半月板。半月板下面平坦,上面凹陷,外缘薄。内侧半月板较大,呈“C”形,前窄后宽,外侧半月板较小,近似“O”形,外缘与关节囊相连[4-6]。半月板有独特的供血特点,一般把半月板分为3个区:①红区,半月板边缘1/3区为边缘血运区,有良好的愈合能力。②红白区,半月板中1/3区为边缘血运区,有潜在的愈合能力。③白区,半月板内侧1/3区为无血运区,愈合能力较差[11]。 研究证明,半月板传送负荷随着膝关节屈曲和旋转的不同程度而变化,膝关节伸直时,压力负荷的50%通过半月板来传递扩散。运动时,半月板承受了人体70%的负重区域,降低了胫骨的受力,对膝关节中的软骨发挥保护作用。如果将半月板切除,则胫骨平台上的峰压力可上升2倍,并将引起软骨退变[12]。膝关节屈曲时半月板承受的压力负荷则达80%-90%。正常膝关节吸收震荡的能力比半月板切除后的膝关节要高出20%。半月板中的水分占其全部湿重的74%。在压力之下,液体可自由流动并被挤入关节间隙,对膝关节起润滑作用。 半月板是膝关节复杂的生物力学结构所必需的组成部分。半月板随着胫骨一起运动,内侧半月板较外侧半月板移位小,且半月板在膝关节伸屈过程中可以变形以适应膝关节的解剖形态,保持了膝关节几何形态的协调,从而维持了膝关节的运动协调性。 2.2 膝关节半月板的损伤机制 半月板的损伤与“半月板的矛盾运动”有关[13],当运动过程中,如果运动者在违反人体生理结构状态下完成技术动作,或者突然性做出不符合惯性力的动作,增加了半月板的承重负荷,超过了其能够承受的压力,导致半月板损伤。当膝关节屈曲伴有扭转或旋转动作时,半月板自身产生矛盾运动,在股骨和胫骨平台之间产生剧烈的研磨从而引起半月板损伤[4-5,7]。当膝关节屈曲,如果小腿外展外旋,大腿突然内收内旋并伸直关节,导致内侧半月板损伤;如果小腿内收内旋,大腿突然外展外旋并伸直关节,导致外侧半月板损伤[9-10]。半月板的损伤分为边缘型撕裂、前角撕裂、后角撕裂、横行撕裂以及水平撕裂等,因损伤机制不同而产生不同类型的撕裂损伤。 2.3 生物材料修复半月板损伤 研究证明,半月板损伤时,部分半月板切除治疗的效果优于全部切除治疗的效果,对于半月板需要全部切除的患者,应给予半月板代替物置入治疗,使膝关节的稳定性能够继续维持。戴力扬[14]对行半月板全切的36名青少年作了远期随访,随访时间为10-33年,平均15.2年,发现有84%的膝关节发生退行性改变。无论半月板全切除或半月板部分切除,关节软骨特别是胫骨均有不同程度的退变。关节镜下行半月板切除比开放式半月板切除创伤小,但同样不能避免由于关节内应力增大而导致的关节软骨变性、疲劳骨折以及骨性关节炎等并发症的发生。且关节镜操作技术复杂,以及容易导致上述并发症的发生,使关节镜在半月板损伤治疗中的应用受限[15]。由于半月板损伤后切除治疗存在较大的缺陷,很多学者建议采用半月板缝合治疗,对发生在边缘25% 以内区域的撕裂,前后附着点完整者进行切开修补缝合或在关节镜下缝合。可使部分边缘型损伤获得愈合,但对单纯无血运区的损伤无治愈效果,缝合后仍遗留并发症。为解决半月板治疗中存在的各种问题,人们对此进行了深入研究,组织工程材料的应用成为治疗半月板损伤的主要方法。 1977年,Green首次打开软骨组织工程学研究领域的大门,为解决半月板移植术中存在的困难,最初人们应用聚羟乙酸、聚乳酸、碳素纤维聚合物等人工合成材料,制作人工半月板,这种人工合成的半月板仅起填充支撑作用,柔软性、蠕变性及生理功能难以达到人体自身半月板的要求。随着组织工程学的发展和组织工程材料的不断成熟,组织工程学的治疗技术也开始应用于运动医学领域治疗半月板损伤。 2.3.1 天然生物支架材料修复半月板损伤 生物支架材料能够为软骨细胞提供生长所需要空间环境,调节生物活性因子与细胞之间的相互作用,促进细胞附着、分化以及细胞表面因子受体表达[5-9]。理想的生物支架材料应该具有良好的生物相容性、可降解性以及充分的孔隙结构和承载生长因子的能力等[16-17]。根据生物支架材料的来源分为天然生物材料和人工合成高分子材料。天然生物材料包括纤维蛋白、透明质酸和胶原等。透明质酸是一种独特的线性大分子粘多糖,由葡萄糖醛酸和N-乙酰氨基葡萄糖的双糖单位反复交替连接而成。透明质酸广泛存在于动物和人体结缔组织细胞间质中,在眼玻璃体、皮肤、脐带、软骨和关节滑液中具有较高的含量,血清中的含量最低。近年来透明质酸作为一种可吸收可降解生物材料,因其高度的粘弹性、可塑性、渗透性和良好的生物相容性,作为药用材料在医药领域的应用中取得显著进展[18]。 透明质酸是构成关节和滑液的主要成分,当发生骨性关节炎、类风湿性关节炎以及其它感染性和非感染性关节疾病时,透明质酸在关节内的产生和代谢即发生异常,滑液中透明质酸的浓度和相对分子质量明显降低,软骨发生降解和破坏,导致关节生理功能障碍。Mendoza等[19]用Cu2+与过氧化氢反应模拟芬顿(Fenton)反应,称为仿Fenton反应,进一步验证了骨性关节炎、类风湿性关节炎均与透明质酸的降解有关,黏弹性补充疗法由此应运而生,旨在通过补充外源性透明质酸,恢复滑液的润滑功能。迄今为止,大量的临床应用结果表明,透明质酸对骨性关节炎、类风湿性关节炎等关节疾病的治疗效果明确,安全,具有很好的应用前景。例如,关节镜微骨折技术用于治疗关节软骨损伤可以促进骨髓干细胞生长,刺激软骨再生,但是这种再生软骨是纤维软骨,不是透明软骨,而且要薄于正常软骨。Kang等[20]在采用关节镜微骨折技术治疗兔关节软骨损伤的基础上应用透明质酸凝胶,组织学分析结果显示,再生软骨的厚度明显大于无透明质酸凝胶组,且更接近于透明软骨,表明透明质酸可明显促进骨折愈合。 胶原半月板支架材料也是研究较为成熟的半月板替代物,未交联的胶原支架材料较化学交联后的支架材料具有更好的生物相容性,植入半月板缺损部位后可吸引关节内的细胞浸润生长,最终可以形成纤维软骨组织[21-22]。由Ⅰ型胶原、硫酸软骨素和透明质酸复合生成的胶原半月板植入物,已应用于临床,胶原半月板植入物表现出良好的治疗效果,形成了纤维软骨组织,明显改善患者膝关节症状,延缓了关节软骨的退行性病变[23]。 2.3.2 人工合成高分子生物支架材料修复半月板损伤 除了天然生物材料之外,许多学者对人工合成的高分子聚合物生物材料作为组织工程化纤维软骨的支架材料也进行了深入的研究,其中研究最多的是聚羟基乙酸和聚乳酸。聚羟基乙酸具有良好的生物相容性和生物可降解性,其降解产物羟乙酸对细胞无毒副作用,可最终代谢为水和二氧化碳。聚羟基乙酸纤维可以通过聚乳酸的有机溶剂塑形,提高支架材料的力学性能[24]。聚羟基乙酸的孔隙率较大,而且降解速度可人为控制,是研究纤维软骨细胞在三维条件下的生物学行为良好的载体[25-26]。为了提高材料的生物活性和力学性能,越来越多的研究关注于天然材料和人工合成材料复合生物材料的研究,天然材料具有良好的生物相容性,但力学强度不能达到生理半月板的要求,而人工合成材料往往具有良好的机械性能,但是其生物活性不高,复合材料就是利用二者的优点,形成既具有一定力学强度又具有良好生物活性的支架材料。 2.4 SCI数据库2000至2013年收录生物材料修复半月板损伤的相关研究文献举例 SCI数据库2000至2013年收录生物材料修复半月板损伤的相关研究文献中,被引频次较高的文献举例见表1。"
| [1] Smillie IS. The congenital discoid meniscus. J Bone Joint Surg Br. 1948;30B(4):671-682.
[2] 祝云利.纤维软骨细胞-胶原复合物修复半月板损伤的实验研究[D].上海:第二军医大学,2001:1-70.
[3] 中国知网.中国学术期刊总库[DB/OL]. 2013-11-10. https://www.cnki.net
[4] 徐爱生,卓士雄.半月板的生物力学及其损伤修复的研究进展[J].内蒙古中医药,2009,28(1):85-88.
[5] 裴彩利.半月板运动损伤的组织工程修复[J].中国组织工程研究与临床康复,2011,15(46):8718-8721.
[6] 林勇,潘树栋.半月板运动损伤修复中组织工程材料的应用[J].中国组织工程研究与临床康复,2010,14(12):2237-2240.
[7] 李晓艳.生物材料修复半月板运动损伤[J].中国组织工程研究与临床康复,2011,15(8):1487-1490.
[8] 钱赫亮.运动性半月板损伤及组织工程半月板的应用[J].中国组织工程研究与临床康复,2011,15(8):1487-1490.
[9] 葛廷云.组织工程材料修复半月板运动损伤的可行性及特点[J].中国组织工程研究与临床康复,2011,15(8):1479-1482.
[10] 郑学美.膝关节损伤修复后的生物力学分析[J].中国组织工程研究,2012,16(21):3987-3990.
[11] 胡慧容.半月板的功能及损伤后治疗的研究进展[J].科海故事博览•科教创新,2010,(11):175-176.
[12] Fukubayashi T, Kurosawa H. The contact area and pressure distribution pattern of the knee. A study of normal and osteoarthrotic knee joints. Acta Orthop Scand. 1980;51(6): 871-879.
[13] 李峰.专项运动员半月板损伤的生理学及生物力学分析与预防对策[J].福建体育科技,2008,27(4):41-44.
[14] 戴力扬,张文林,周志华,等.青少年半月板切除术的远期疗效[J].中国运动医学杂志,1996,15(1):48-50.
[15] 张帅明,高顺生,张东龙.膝关节半月板损伤的修复及临床治疗研究进展[J].北京体育师范学院学报,2000,12(2):82-85.
[16] 鄂征,刘流.医学组织工程技术与临床运用[M].北京:北京出版社, 2003:378-379.
[17] 林智军,王万明.组织工程软骨生物支架材料研究新进展[J].中国组织工程研究与临床康复,2007,11(18):3625-3628.
[18] 王超,张明春.透明质酸的制备及作为药用材料方面的应用进展[J].中国医药生物技术,2009,4(6):452-454.
[19] Mendoza G, Alvarez AI, Pulido MM, et al. Inhibitory effects of different antioxidants on hyaluronan depolymerization. Carbohydr Res. 2007;342(1):96-102.
[20] Kang SW, Bada LP, Kang CS, et al. Articular cartilage regeneration with microfracture and hyaluronic acid. Biotechnol Lett. 2008;30(3):435-439.
[21] Masur SK, Dewal HS, Dinh TT, et al. Myofibroblasts differentiate from fibroblasts when plated at low density. Proc Natl Acad Sci USA. 1996;93(9):4219-4223.
[22] Zhang YQ, Zhang WJ, Liu W, et al. Tissue engineering of corneal stromal layer with dermal fibroblasts: phenotypic and functional switch of differentiated cells in cornea. Tissue Eng Part A. 2008;14(2):295-303.
[23] Gastel JA, Muirhead WR, Lifrak JT, et al. Meniscal tissue regeneration using a collagenous biomaterial derived from porcine small intestine submucosa. Arthroscopy. 2001;17(2): 151-159.
[24] Stewart AA, Byron CR, Pondenis HC, et al. Effect of dexamethasone supplementation on chondrogenesis of equine mesenchymal stem cells. Am J Vet Res. 2008;69(8): 1013-1021.
[25] Steadman JR, Rodkey WG. Tissue-engineered collagen meniscus implants: 5- to 6-year feasibility study results. Arthroscopy. 2005;21(5):515-525.
[26] Aufderheide AC, Athanasiou KA. Comparison of scaffolds and culture conditions for tissue engineering of the knee meniscus. Tissue Eng. 2005;11(7-8):1095-1104.
[27] Baker BM, Mauck RL. The effect of nanofiber alignment on the maturation of engineered meniscus constructs. Biomaterials. 2007;28(11):1967-1977.
[28] Buma P, Ramrattan NN, van Tienen TG, et al. Tissue engineering of the meniscus. Biomaterials. 2004;25(9): 1523-1532.
[29] Hidaka C, Ibarra C, Hannafin JA, et al. Formation of vascularized meniscal tissue by combining gene therapy with tissue engineering. Tissue Eng. 2002;8(1):93-105.
[30] Baker BM, Nathan AS, Huffman GR, et al. Tissue engineering with meniscus cells derived from surgical debris. Osteoarthritis Cartilage. 2009;17(3):336-345.
[31] Pabbruwe MB, Kafienah W, Tarlton JF, et al. Repair of meniscal cartilage white zone tears using a stem cell/collagen-scaffold implant. Biomaterials. 2010;31(9): 2583-2591.
[32] Kang SW, Son SM, Lee JS, et al. Regeneration of whole meniscus using meniscal cells and polymer scaffolds in a rabbit total meniscectomy model. J Biomed Mater Res A. 2006;78(3):659-671.
[33] Hoben GM, Athanasiou KA. Meniscal repair with fibrocartilage engineering. Sports Med Arthrosc. 2006;14(3):129-137.
[34] Kobayashi M. A study of polyvinyl alcohol-hydrogel (PVA-H) artificial meniscus in vivo. Biomed Mater Eng. 2004;14(4): 505-515.
[35] Ode GE, Van Thiel GS, McArthur SA, et al. Effects of serial sectioning and repair of radial tears in the lateral meniscus. Am J Sports Med. 2012;40(8):1863-1870.
[36] Anz AW, Rodkey WG. Biological enhancement of meniscus repair and replacement. Sports Med Arthrosc. 2012;20(2): 115-120.
[37] Tan Y, Zhang Y, Pei M. Meniscus reconstruction through coculturing meniscus cells with synovium-derived stem cells on small intestine submucosa-a pilot study to engineer meniscus tissue constructs. Tissue Eng Part A. 2010;16(1): 67-79.
[38] Gruber HE, Mauerhan D, Chow Y, et al. Three-dimensional culture of human meniscal cells: extracellular matrix and proteoglycan production. BMC Biotechnol. 2008;8:54.
[39] Martinek V, Ueblacker P, Bräun K, et al. Second generation of meniscus transplantation: in-vivo study with tissue engineered meniscus replacement. Arch Orthop Trauma Surg. 2006; 126(4): 228-234.
[40] Gastel JA, Muirhead WR, Lifrak JT, et al. Meniscal tissue regeneration using a collagenous biomaterial derived from porcine small intestine submucosa. Arthroscopy. 2001; 17(2): 151-159.
[41] Kobayashi M, Chang YS, Oka M. A two year in vivo study of polyvinyl alcohol-hydrogel (PVA-H) artificial meniscus. Biomaterials. 2005;26(16):3243-3248.
[42] Aufderheide AC, Athanasiou KA. Assessment of a bovine co-culture, scaffold-free method for growing meniscus-shaped constructs. Tissue Eng. 2007;13(9): 2195-2205.
[43] Arnoczky SP,Warren RF,Hennerbichler D,et al.Repair response of the inner and outer regionsof the porcinemeniscus invitro.Am J Sports Med. 2007;35(5): 754-762.
[44] Albrecht-Olsen P,Kristensen G,Burgaard P,et al.Theregenera-tive effectsofplatelet rich plasma onmeniscalcells invitro and its in vivo application with biodegradable gelatinhydrogel.Tissue Eng. 2007; 13(5): 1103-1112.
[45] Metcalf MH,Barrett GR. Prospective evaluation of1 485 meniscal tearspatterns in patientswith stable knees.Am J Sports Med.2004;32(3):675-680.
[46] 宁廷民,孙磊.半月板损伤的治疗[J].中国矫形外科杂志, 2008, 16(14):1084-1087.
[47] 郭美娜,张喜才.早期康复治疗对膝关节损伤患者术后疗效的影响[J].中华物理医学与康复杂志,2008,30(9):644-645.
[48] 白波,赵景良.半月板损伤修复和移植术的研究进展[J].中国误诊学杂志,2009,9(23):5559-5560.
[49] Kurzwell PR,FriedmanMJ.M eniscus: Resection,repair,and replacement. Arthroscopy.2002;18(2):33.
[50] KobayashiM.A study of polyvinyl alcohol2hydrogel(PVA-H) artificialmeniscus in viv. Bioned Mater Eng.2004;14(4):505.
[51] Shieh A, Bastrom T, Roocroft J, et al. Meniscus tear patterns in relation to skeletal immaturity: children versus adolescents. Am J Sports Med. 2013 Dec;41(12):2779-83.
[52] Xu C, Zhao J. A meta-analysis comparing meniscal repair with meniscectomy in the treatment of meniscal tears: the more meniscus, the better outcome? Knee Surg Sports Traumatol Arthrosc. 2013 May 14.
[53] Di Matteo B, Tarabella V, Filardo G, et al. Thomas Annandale: the first meniscus repair. Knee Surg Sports Traumatol Arthrosc. 2013;21(9):1963-1966.
[54] Taylor SA, Rodeo SA. Augmentation techniques for isolated meniscal tears. Curr Rev Musculoskelet Med. 2013;6(2): 95-101.
[55] Edouard P, Rannou F, Coudeyre E. Animal evidence for hyaluronic acid efficacy in knee trauma injuries. Review of animal-model studies. Phys Ther Sport. 2013;14(2):116-123.
[56] Lee YH, Nyland J, Burden R, et al. Repair of peripheral vertical meniscus lesions in porcine menisci: in vitro biomechanical testing of 3 different meniscus repair devices. Am J Sports Med. 2013;41(5):1074-1081.
[57] Schillhammer CK, Werner FW, Scuderi MG, et al. Repair of lateral meniscus posterior horn detachment lesions: a biomechanical evaluation. Am J Sports Med. 2012;40(11): 2604-2609. |
| [1] | Li Jing, Yang Long, Wang Jian-ji, Liu Qin, Zou Qiang, Sun Yu, Ma Min-xian, Ye Chuan. Three-dimensional reconstruction based on DICOM data and its application for orthopedic implants [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 1046-1051. |
| [2] | Lu Zhong-lin, Cao Zhi-qiang, Gao Guo-liang, Jing Qing-ling, Zhang Wei, Huang Yong. Incidence and risk factors of deep vein thrombosis during waiting period before operation for calcaneal fractures by ultrasound elastography [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(3): 423-427. |
| [3] | Bai Zhao-hui, Zhang Ying, Yin Qing-shui, Xia Hong, Wang Jian-hua, Xu Jun-jie. Navigational template applied in the orthopaedic field in China: a bibliometric analysis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(19): 3023-3030. |
| [4] | Yang Min, Ma Xiang-yang, Yang Jin-cheng, Chen Shu-jin, Zou Xiao-bao. Biomechanical properties of a novel automatic anti-rotation posterior atlantoaxial internal fixation system: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(19): 3031-3037. |
| [5] | Qiu Hao, Lu Min-peng, Dong Jing, Zhang Zhong-zu, Chu Tong-wei, Wang Qun-bo, Quan Zheng-xue, Jiang Dian-ming. Subtotal corpectomy and reconstruction with titanium mesh cage implantation and pedicle screw fixation through posterior approach in treatment of thoracolumbar burst fracture or thoracolumbar fracture dislocation [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(53): 7932-7938. |
| [6] | Zha Yuan-yu, Yang Yang, Zhou Yi-chi, Wei Ren-xiong, Zhang Shu-wei, Jin Wei. Meta-analysis of anterior screw fixation versus posterior cervical fusion in treatment of odontoid fractures [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(48): 7288-7296. |
| [7] | Xu Jian-da, Gao Yi, Zhao Hong, Peng Li-bo, Zheng Chong, Wang Bin, Qu Yu-xing, Zhao Jian-ning. Viper system and pedicle screw fixation for treating thoracolumbar compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6583-6589. |
| [8] | Jiang Zi-wei, Huang Feng, Zheng Xiao-hui, Hu Qun-sheng, Pang Zhi-hui, Zhou Guang-quan, Li Yue . Different fixations for intertrochanteric fracture affect proximal femur stress: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6599-6605. |
| [9] | Xu Can, Li Ming-qing, Wang Cheng-gong, Li Kang-hua, Liu Hua. Research progress of bone microarchitecture and microdamage detection [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6673-6681. |
| [10] | Liu Ya-pu, Hou Xiu-wei, Wu Guang-liang, Xia Hong. Comparison of stress distribution of adjacent segments after artificial cervical disc replacement versus anterior cervical discectomy and fusion: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6541-6548. |
| [11] | Ma Liang, Guo Wei-chun, Xu Yong-tao. Single iliac screw and dual iliac screws and titanium mesh cage fixation in the reconstruction of lumbosacral defects with finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(39): 5859-5866. |
| [12] | Zhang Hai-feng, Yin Ai-hua, Dong Yi, Song Cui-rong, Liu Yuan-yuan, Ren Guo-shan, Pang Yin. Finite element method for analyzing the stress distribution of acetabulum under different loads [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(39): 5867-5872. |
| [13] | Xiong Yang, Yu Xing. Research progress of biomechanics and finite element analysis of lumbar interspinous devices [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(39): 5919-5928. |
| [14] | Zhang Yi-long, Ren Lei, Sun Zhi-jie, Wang Ya-hui, Sun He. New vertebral compression fractures after vertebroplasty: association with osteoporosis and spinal sagittal imbalances [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(35): 5263-5269. |
| [15] | Shang Hong-tao, Wang Quan, Liu Bin, Liu Wei. Biomechanical characteristics of external fixation for tibial plateau fractures [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(31): 4651-4657. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||